Improving Parkinson's gait & balance through selective stimulation

Deep brain stimulation (DBS) is an effective treatment for many Parkinson's disease (PD) symptoms, but results vary, with gait & balance being among the least responsive symptoms. It's agreed that effectiveness of DBS in PD depends on the location of stimulation, but there's less agreement on where the best location is, especially regarding the best location for improving gait and balance. With colleagues in the Center for Magnetic Resonance Research (CMRR) and Biomedical Engineering we are relating DBS effects on gait to stimulation location, using very detailed, maps of local neuroanatomy. Under a 5-year grant from the National Institutes of Health, we are designing and testing DBS to selectively activate neural pathways that optimally treat Parkinson's gait & balance symptoms.
Selective activation of two different axonal fiber tracts from a single DBS electrode, using ultra high field strength MRI, patient-specific neural models, and nonliner particle swarm optimization.
Time course of Deep Brain Stimulation Effects
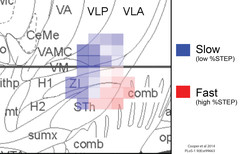
Surprisingly, deep brain stimulation's effects on Parkinson's symptoms are not instantaneous: they wash in/out gradually when stimulation is turned on/off. For one symptom -- bradykinesia (slowness of movement) -- we found that rate of washout depends on the characteristics of the patient (disease duration) and also on the location of stimulation. This is a clue to the physiological mechanisms by which DBS affects the symptom. Other symptoms wash out differently, which may mean DBS acts on them by different mechanisms. We are studying DBS washout for two other symptoms: gait, and speech.
Percent of fast-vs.-slow-washout DBS effect on bradykinesia varies with location of stimulation
Measuring Parkinsonian Postural Instability
Loss of balance (postural instability) is another of the most troublesome Parkinson's symptoms. A bipedal creature stays upright by dint of constant postural adjustments, sometimes shifting the feet to keep the center of support under the center of mass. That reflex is impaired in PD, and that impairment responds poorly to existing treatments. Little is known about how varying forms of deep brain stimulation affect it. To study that, we are developing a system to study the stepping reflex in response to a standardized balance perturbation.
Volunteer in safety harness, ready for balance perturbation
Neural Recordings During Gait
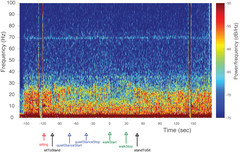
It is difficult to record electrical activity, in human brains, from deep structures such as the basal ganglia. It gets even more difficult during activities such as gait. In collaboration with the University of Minnesota Udall Center, we are using a special implantable DBS device which not only stimulates but also records electrical activity to investigate how neural activity varies in relation to types of gait events, including phase of gait cycle, "freezing of gait" and the sudden appearance of obstacles on the walking path.
Time-frequency plot of globus pallidus local field potential power, as the patient sits in a chair, then stands quietly, then walks, then stands again, then sits down.
Response Inhibition
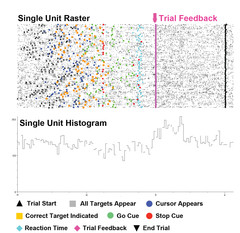
People with Parkinson's not only have difficulty initiating movements; they also have difficulty aborting movements once initiated ("response inhibition, RI"). Impairment of RI may underly "impulse control disorders," seen in Parkinson's and other neurological and psychiatric disorders. Deep brain stimulation, while it improves response initiation, might either improve, or worsen response inhibition. Both effects have been reported, and the variability might reflect variations in the precise location of stimulation. To examine this, we are studying a stop-signal task, requiring inhibition of a prepotent target-capture response, and comparing performance in controls and in Parkinson's patients on and off medications and DBS.
Microelectrode recording during surgery, from a neuron in the subthalamic nucleus of a Parkinson's patient performing the stop-signal task